Abstract
Chlamydia psittaci infection among humans (psittacosis) and pet birds (avian chlamydiosis), also known as parrot disease, parrot fever, and ornithosis, is a zoonotic bacterial disease. Humans most often become infected by inhaling the organism when urine, respiratory secretions, or dried feces of infected birds are dispersed in the air as very fine droplets or dust particles. C. psittaci infection of humans can cause influenza-like symptoms, such as fever of abrupt onset, pronounced headache, and dry cough, and can lead to severe pneumonia and non-respiratory health problems. Infection can also be asymptomatic. There is no vaccine for this infection. The disease is treatable with a tetracycline antibiotic, usually doxycycline, or a second-line therapy such as erythromycin or azithromycin. With appropriate treatment, the infection is rarely fatal. This report describes a case of severe, community-acquired pneumonia possibly due to C. psittaci in a resident of Colorado and examines significant clinical and epidemiological characteristics of psittacosis that affect confirming the diagnosis and managing the risks of exposure to psittacine (parrot-type) birds.
Background
The 24-year-old spouse of an active duty Army service member sought care at the Evans Army Community Hospital (EACH) emergency department (ED) at Fort Carson, CO, several times over the course of 21 days. On 12 Feb. 2018, the patient presented to the ED with fever, gastrointestinal symptoms of diarrhea, vomiting, and cough. Although her gastrointestinal symptoms resolved, the patient returned on 23 Feb. 2018 with persistent cough, fever, chills, lightheadedness, and sinus tachycardia. The patient's chest x-ray was clear, and she was diagnosed with a viral syndrome, not influenza. The patient returned to the ED on 3 March 2018 with dyspnea upon mild exertion (e.g., turning over in bed), occasional wheezing, mild fever (99.3°F), peripheral capillary oxygen saturation (SpO2) of 91% on room air, and lightheadedness. Chest x-ray showed patchy airspace density in the left lung base consistent with early pneumonia, a clear right lung, and no pleural effusion. She was discharged home with a diagnosis of lobar pneumonia with prescriptions for azithromycin 250 mg orally per day for 5 days and the expectorant guaifenesin with codeine. The patient returned to the ED the following day (4 March 2018) with worsening symptoms of dyspnea, fever to 100.7°F, SpO2 of 88% on room air, chest pain, myalgia, pain with breathing, tachycardia, photophobia, and lightheadedness. Chest x-ray revealed an interval increase in the left lower lobeinfiltrate, but the right lung remained clear with no pleural effusion. The patient was diagnosed with pneumonia and was administered a course of levofloxacin 500 mg intravenously with O2 therapy. Later that day, she was transported to a local hospital (UCHealth Memorial Hospital Central of Colorado Springs) and admitted for pneumonia and acute respiratory failure with hypoxia. The patient was hospitalized from 5 March through 7 March 2018. The patient was not admitted to EACH because of a brush fire on Fort Carson at the time.
UCHealth Memorial Hospital Central admission screening questions about pets in the home revealed that the patient owned a cockatiel. The civilian hospital collected a serum specimen on 5 March 2018 for a C. psittaci microimmunofluorescence (MIF) immunoglobulin M (IgM) antibody test. Results received on 10 March 2018 included the following values: C. trachomatis IgG <1:64, C. trachomatis IgM 1:320, C. pneumoniae IgG <1:64, C. pneumoniae IgM 1:640, C. psittaci IgG <1:64, C. psittaci IgM 1:320. The reference range for the IgG values was listed as <1:64 for each test. The reference range for the IgM values was listed as <1:20 for each test.
The MIF test was not complete before discharge, but the patient was treated presumptively for C. psittaci infection with a course of levofloxacin and doxycycline while hospitalized. Tests for influenza A and B virus antigen, other respiratory viruses, and Group A streptococci were negative. On 7 March the patient was considered medically stable and was discharged with a 3-day course of levofloxacin (750 mg/day), a 4-day course of doxycycline (200 mg/day), and an albuterol inhaler. The patient was instructed to follow up with her primary care manager in 7 to 10 days.
After UCHealth Memorial Hospital of Colorado Springs reported the results of the MIF test to the Colorado Department of Public Health and Environment (CDPHE), the CDPHE state zoonosis veterinarian called the Fort Carson Department of Public Health on 16 March 2018 to report the case and to recommend an interview with the patient and follow-up on her bird. Army Public Health Nursing (APHN) personnel interviewed the patient, provided health education, and advised the patient on treatment options for her bird based on recommendations from the Fort Carson veterinarian. Specific cleaning and disinfecting instructions were provided via a document created by the National Association of State Public Health Veterinarians titled, "Psittacosis and Avian Chlamydiosis Checklist for Owners of Infected Birds." 1 The patient owned 1 bird, a cockatiel, species Nymphicus hollandicus. The patient reported that this was the only bird in the home and that it had been purchased as a chick approximately 3 years previously from a breeder in California. Based on the patient's report, the bird in her home was the most likely exposure. The patient did not work or volunteer at a pet store or shelter and had not visited other homes that housed birds. The bird had not appeared to be sick.
APHN personnel provided the patient, ED personnel, and the Fort Carson veterinarian with educational material referencing the Compendium of Measures to Control Chlamydia psittaci Infection Among Humans (Psittacosis) and Pet Birds (Avian Chlamydiosis), 2017” (hereafter referred to as "the compendium").2 Two weeks posthospitalization, on 23 March 2018, the patient was seen again at the ED with pain in her left lung, cough, and dyspnea. Her chest x-ray was clear. She was diagnosed with a recurrence of psittacosis pneumonia and was treated with intravenous doxycycline and dexamethasone. The patient noted that this treatment did not result in a significant improvement in her symptoms, so 5 days later, she was given a second 14-day course of oral doxycycline 200 mg/day because her bird had yet to be examined and treated for psittacosis and because her bird could potentially be shedding the bacteria even though the bird did not appear sick. The patient's husband was evaluated at the ED because of similar symptoms, but no significant diagnosis was made. No family members or other contacts were ill. The patient denied any recent travel. Convalescent testing was performed. Serum for the MIF antibody test was collected at the EACH lab on 2 April 2018. Results available on 5 April 2018 showed that the patient's serological test results specific for C. psittaci were negative (IgM <1:16 and IgG <1:10). The laboratory that performed the test during the acute phase of the illness was different from the lab that performed the convalescent test.
APHN personnel consulted with the Fort Carson veterinary clinic, which provided the name of an avian specialist veterinarian in Colorado Springs and recommended the bird be evaluated by that specialist, as military veterinary clinics do not provide clinical care to exotic animals, including avian species. However, the patient took the bird to her regular local veterinarian, an off-post civilian veterinarian, instead of the recommended local civilian avian specialist veterinarian. On 20 March 2018, Chlamydia DNA testing was carried out on a choanal/cloacal swab specimen from the bird. On 28 March 2018, the patient's local civilian veterinarian informed her that the DNA test results on her bird's specimen were negative. Because the bird was asymptomatic and the only bird being tested, the recommendations in Appendix 1 of the compendium were indicated. These recommendations state that "diagnosis of avian chlamydiosis can be difficult, especially in the absence of clinical signs. A single testing method might not be adequate. Therefore, use of a combination of culture, polymerase chain reaction (PCR)-based detection, and antibody detection is recommended, particularly when only 1 bird is tested."1 APHN personnel had initially provided the local civilian veterinary office with the full compendium and sent a highlighted copy of just Appendix 1 after the DNA test results came back negative. Despite having received a copy of the testing protocol for C. psittaci in birds,1 the local veterinarian advised against the recommended testing procedure, stating that the bird appeared well (veterinarian, phone call, March 2018). APHN personnel called the state zoonosis veterinarian again to discuss the issue. The state veterinarian offered to speak to the local veterinarian directly and asked the APHN to convey that offer. Multiple attempts by the APHN to speak to the local civilian veterinarian were unsuccessful. At that point, the state veterinarian and APHN agreed that enough attempts had been made to collaborate with the local treating veterinarian. The patient did not have the funds to have the bird tested elsewhere, so the bird was neither fully tested nor treated.
The Fort Carson Department of Environmental Health (EH) was consulted to discuss the ramifications of an infected bird located in on-post housing. EH recommended that the bird's owners clean the home as indicated in "Part III. Recommendations for Controlling Infection Among Humans and Birds," 2 replace the carpeting in the home, and keep the birdcage on a non-porous surface that could be more easily disinfected. These recommendations were made for the patient's asymptomatic bird, which was believed to be associated with a human case of psittacosis before the bird was treated. The house was carpeted and the housing office stated the occupants would have to pay out of pocket to have the flooring replaced. The patient and her husband did not have the funds to have the flooring replaced in their rental home. The patient's husband vacuumed the area around the birdcage while the patient was being treated for and recovering from pneumonia. APHN personnel informed the patient and her husband to use the vacuum cleaner with caution because vacuuming may aerosolize infectious C. psittaci particles, as noted in the compendium.2 The patient's condition improved over the course of the next few months, although she continued to have a chronic cough. The patient revisited the ED in June and July 2018 with symptoms of cough, lightheadedness, and nosebleeds that resulted in a diagnosis of allergic rhinitis and epistaxis.
Editorial Comment
Avian chlamydiosis, called psittacosis when it occurs in humans, is a zoonotic disease caused by the obligate intracellular gram-negative bacterial pathogen called C. psittaci, which is distinct antigenically and genetically from other Chlamydia species.3 Because several diseases affecting humans can be caused by other species of Chlamydia, the disease resulting from the infection of humans with C. psittaci frequently is referred to as psittacosis rather than chlamydia.
Psittacosis has a worldwide distribution and can occur sporadically or in epidemic fashion at any time of the year.3 In the U.S., from 2003 through 2014, 112 psittacosis cases were reported to the Centers for Disease Control and Prevention (CDC) through the Nationally Notifiable Diseases Surveillance System.2 This number is likely an underestimate of the actual number of cases because psittacosis is difficult to diagnose. The disease is also known as parrot disease, parrot fever, and ornithosis because most psittacosis cases result from exposure to infected pet birds, particularly the Psittaciformes, the order of birds that includes parrots, macaws, cockatiels, and parakeets.2,4–7 However, psittacosis is prevalent in poultry, pet birds, and wild birds and causes economic losses to the poultry industry and the pet trade.8,9
Humans most often become infected by inhaling the organism when urine, respiratory secretions, or dried feces of infected birds are dispersed in the air as very fine droplets or dust particles. Once a bird is infected, the feathers and feces may be contagious many months after the acute illness has resolved.10 Other sources of exposure include mouth-to-beak contact, a bite from an infected bird, the handling of infected birds's plumage or tissues, and the dissection of dead birds or evisceration in slaughterhouses. Even short-term exposures can lead to symptomatic infection11,12; therefore, some patients with psittacosis may not recall or report having any contact with birds.2
The symptoms and severity of psittacosis can vary significantly. Some individuals are asymptomatic or only have a very mild infection; others can develop serious widespread infection that affects other parts of the body. The onset of symptoms can be characterized as occurring suddenly or insidiously. C. psittaci infection of humans most commonly presents in young or middle-aged adults as fever of abrupt onset, pronounced headache, and dry cough.2 Affected individuals also may develop chills, myalgia, and malaise. Pneumonia, which is often evident on chest x-ray, commonly occurs and can be severe. Pulse–temperature dissociation (fever without elevated pulse), enlarged spleen, and rash are sometimes observed and suggest a diagnosis of psittacosis for patients with community-acquired pneumonia. Auscultatory findings may underestimate the extent of pulmonary involvement; also, radiographic findings may include lobar or interstitial infiltrates.2 Frequent epistaxis and hepatomegaly are also familiar occurrences. Although the lungs are the organ most often affected by psittacosis, the disease can potentially affect many organ systems in the body, including the gastrointestinal tract, heart, liver, skin, and central nervous system. The incubation period is usually between 7 and 14 days but can be as long as 39 days.4
The differential diagnosis of psittacosis is wide ranging but can be limited based on the specific clinical presentation of the patient. For a patient with atypical pneumonia, the other etiologies to consider include C. pneumoniae, Mycoplasma pneumoniae, and Legionella pneumophila.2,13 If the affected patient primarily has a febrile illness without any localizing signs, then, besides psittacosis, other systemic conditions such as influenza, endocarditis, myocarditis, septicemia, vasculitis, Coxiella burnetii infection, leptospirosis, and brucellosis should also be considered.2,14 When extrapulmonary manifestations predominate, the patient should be evaluated for the causes of the most prominent manifestation, such as gastroenteritis, hepatitis, meningitis, or encephalitis.2,14 Infection with C. psittaci has been reported to affect organ systems other than the respiratory tract, resulting in conditions including endocarditis, myocarditis, hepatitis, arthritis, keratoconjunctivitis, encephalitis, and ocular adnexal lymphoma.15
CDC and the Council of State and Territorial Epidemiologists have recognized national case definitions for epidemiologic surveillance of psittacosis that health care providers can use as part of their clinical evaluation in ascertaining a diagnosis of psittacosis.13 It is important for health care providers to use these case classifications as an element in the overall analysis for confirming a clinical diagnosis or determining medical management (Table 1).13 Because psittacosis in humans is a nationally notifiable disease, the CDPHE zoonosis veterinarian called the EACH preventive medicine department to report the case and recommend follow-up care and treatment for the patient and her pet bird.
Diagnosis of psittacosis can be challenging. For example, because the patient's initial test IgM titer results appeared to be positive for all 3 types of Chlamydia (C. trachomatis IgM 1:320; C. pneumoniae IgM 1:640; and C. psittaci IgM 1:320), the definitive diagnosis was open to several interpretations. Specificity challenges with the 3 types of Chlamydia could lead the clinician to the plausible interpretation that the patient had either an infection with C. pneumoniae with cross-reactive antibodies to C. psittaci or an infection with C. psittaci with cross-reactive antibodies to C. pneumoniae. Because the medications for both types of pneumonia are similar, the patient's response to the treatments given multiple times does not clarify the diagnosis. The reported convalescent serological results do not make the diagnosis any clearer either. Also, because acute and convalescent sera should be analyzed simultaneously at the same laboratory,2 the reliability of serological results was diminished, as 2 different labs performed the acute and convalescent tests for the patient. While the presence of the cockatiel in the household suggested a possible diagnosis of psittacosis, it is conceivable that this patient may have had community-acquired pneumonia due to C. pneumoniae and she, by chance, happened to own a cockatiel. It is entirely possible that the cockatiel was not infected with C. psittaci, as demonstrated by its apparent health and the set of negative test results. On the other hand, a single testing method on the bird might not be adequate, and the use of a combination of culture, PCR-based detection, and antibody detection is recommended, particularly when only 1 bird is tested.2 A diagnosis of psittacosis could have been made or ruled out if the cockatiel was examined and tested properly by an avian specialist veterinarian because a confirmed psittacosis diagnosis on the bird would have indicated a more definitive source of exposure for the patient. In this analysis, with no other substantial laboratory evidence to narrow down the diagnosis to 1 of these 2 Chlamydia species (C. pneumoniae or C. psittaci), it appears questionable to definitively diagnose the patient as having psittacosis.
The best method to confirm C. psittaci infection in the patient is serologic testing, namely a MIF antibody test.2 Ideally, most diagnoses are determined by clinical presentation and positive antibodies against C. psittaci in paired sera using MIF methods. Convalescent serum testing for MIF was collected at the EACH lab on 2 April 2018. Results available on 5 April 2018 showed that the patient's serological test results were IgM <1:16 and IgG <1:10. While the MIF antibody test is generally more sensitive and specific than complement fixation tests,16 cross-reactivity with other chlamydiae (C. pneumoniae, C. trachomatis, and C. felis) may occur. For this reason, the compendium recommends that a titer less than 1:128 should be interpreted with caution, and true acute (obtained as close to the onset of symptoms) and convalescent (ideally taken 2–4 weeks later) specimen tests are required for accurate interpretation.2 Because antimicrobial treatment can postpone or weaken the antibody response, a third serum sample 4–6 weeks after the acute sample is recommended.2 Although serologic testing is more commonly used and available than molecular testing, results can often be unclear, subjective in their interpretation, and ambiguous because of intrinsic limitations of this methodology. If feasible, serology should be considered a supportive test that augments the findings of other more dependable assays, such as nucleic acid–based tests.17 Information about laboratory testing is available from state public health departments and the compendium.2 PCR-based testing and assistance can be requested via CDC's Respiratory Diseases Division, and assistance with case investigation can be requested via the U.S. Army Public Health Center and/or the U.S. Army Public Health Command Central.
Tetracycline antibiotics are the drug of choice for treating C. psittaci infection in humans.18 The first choice of therapy for treating mild-to-moderate illnesses is doxycycline 100 mg administered orally twice daily or tetracycline 500 mg 4 times daily for at least 10 to 14 days to prevent relapse. Severely ill patients typically require treatment with intravenous doxycycline hyclate at 4.4 mg/kg/day, divided into 2 infusions per day. Most C. psittaci infections are responsive to antibiotics within 1–2 days; however, relapses can occur. Erythromycin is less effective than doxycycline but can be used in cases where tetracyclines are contraindicated (e.g., tetracycline allergy, during pregnancy, for children). The prognosis for treated psittacosis is excellent, with a mortality rate of less than 1%.19 As has been recommended for the treatment of Rocky Mountain spotted fever, if the benefits outweigh the risks (particularly if the alternative medicine is not effective and it is a life-threatening situation), a tetracycline, such as doxycycline, could be considered in children.20 Prophylactic antibiotics are not routinely administered after a suspected exposure to C. psittaci but may be considered in some circumstances.20
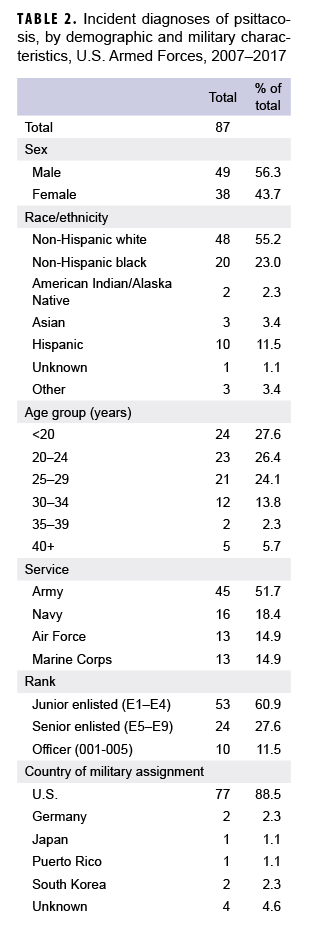
According to data from the Armed Forces Health Surveillance Branch (AFHSB), 87 cases of C. psittaci infection were reported in U.S. service members between 1 Jan. 2007 and 31 Dec. 2017 (AFHSB, unpublished data, 2018) (Table 2). Over half (n=48) of the total C. psittaci cases occurred among non-Hispanic whites. The majority of cases (n=77) occurred in the continental U.S. Slightly more than half of the cases occurred among Army service members (n=45); far fewer cases were diagnosed in members of the Navy (n=16), Air Force (n=13), and Marine Corps (n=13). Of the total cases, 49 were male (56.3%) and 38 were female (43.7%). In addition, seven-eighths (87.4%) of the cases were active duty service members and the remaining cases were Reserve or National Guard members.
When C. psittaci is recognized in a beneficiary of the Military Health System, the case should be promptly reported to local civilian and military public health authorities as soon as laboratory and clinical information are available. Although it is not a requirement to report C. psittaci infection in the Disease Reporting System internet for surveillance purposes per the Armed Forces Reportable Medical Events Guidelines and Case Definitions, 2017,21& he condition can be reported in this system under "Any Other Unusual Condition Not Listed", with "C. psittaci infection" entered in the comment field along with a psittacine bird exposure history and other pertinent information. The number of diagnosed and reported human infections likely underestimates the actual (true) number of cases because psittacosis is difficult to diagnose, is treatable with antimicrobials (which may be employed empirically for therapy of community-acquired pneumonia), and often is not reported. Timely, accurate reporting of probable, suspected, or confirmed cases ensures proper identification, treatment, control, and follow-up of cases.
This possible case of psittacosis presented some significant challenges in that recommended avian testing was not performed and there were confounding circumstances that potentially impacted the health of the patient and her bird. Moreover, this case highlighted some issues related to privatized housing and health concerns that can have a public health impact on military installations. The fact that the patient lived in on-post privatized housing posed a barrier to best public health practice. Despite the strong recommendations from Fort Carson EH, the private housing company refused to replace the carpeting/flooring of the patient's home unless paid for by the patient. The financial limitations of this patient, who is the spouse of a junior enlisted soldier, further impacted the ability to disinfect their home environment. The Fort Carson Department of Public Health may have had more authority if on-post housing were owned by the government. Financial limitations also impeded the patient from having her bird properly evaluated by an avian specialist veterinarian even though one was available in town. These factors, along with the lack of adherence to the Fort Carson APHN and EH public health recommendations, potentially left the patient and those that came in contact with her bird at potential risk for infection. It is unclear if the chronic cough the patient reported was related to C. psittaci infection. The costs to have the bird adequately tested and to replace the carpeting in the patient's home were minimal compared to the costs associated with the multiple visits to the ED and the subsequent hospitalization of the patient. Appropriate diagnostic testing and interpretation as well as treatment of the patient's bird, which was the suspected source of her infection, should have been overseen by an experienced avian specialist veterinarian.
This case demonstrated that early identification of the disease can be challenging because of the non-specific clinical signs that occur during an infection with C. psittaci. Nonetheless, a history of recurrent contact with psittacine bird species, along with indicative symptoms, should generate further diagnostics for psittacosis in order to initiate treatment in humans and contact birds as soon as possible. Local military public health authorities should continue to identify cases of C. psittaci infection and report them as soon as laboratory and clinical information are available. Preventive measures include 1) cleaning cages regularly (dampening cages and other contaminated areas with cleaning solution or disinfectant reduces aerosolization) and keeping birds and cages in well-ventilated areas to prevent the accumulation of infectious dust; 2) employing good hygiene, including frequent hand washing, when handling birds, their feces, and their environments; 3) utilizing gloves, coveralls or disposable gowns, disposable caps, protective eyewear (e.g., goggles), and a properly fitted respirator mask (i.e., a pre-shaped mask that molds firmly around the mouth and nose); and 4) following all instructions provided by the treating veterinarian regarding treatment, isolation and quarantine, follow-up testing, and handling of any ill and exposed birds.
Author affiliations: Epidemiology and Disease Surveillance, U.S. Army Public Health Command Central Region, Joint Base San Antonio-Fort Sam Houston, TX (Dr. Stidham); Evans Army Community Hospital (Ms. Richmond-Haygood)
Acknowledgments: The authors thank Dr. Shauna Stahlman, PhD, MPH, and her colleagues at the Armed Forces Surveillance Branch for providing surveillance case data.
Disclaimer: The view(s) expressed herein are those of the author(s) and do not reflect the official policy or position of the U.S. Army Public Health Command Central Region, Evans Army Community Hospital, U.S. Army Medical Department, U.S. Army Office of the Surgeon General, Department of the Army, Department of Defense, or U.S. Government.
References
- National Association of State Public Health Veterinarians. Psittacosis and avian chlamydiosis checklist for owners of infected birds. http://www.nasphv.org/Documents/PsittacosisChecklistOwners2017.pdf. Accessed 25 Jan. 2019.
- Balsamo G, Maxted AM, Midla JW, et al. Compendium of measures to control Chlamydia psittaci infection among humans (psittacosis) and pet birds (avian chlamydosis), 2017. J Avian Med Surg. 2017;31(3):262–282.
- American Academy of Pediatrics. Chlamydia (Chlamydophila) psittaci. In: Pickering LK, ed. Red Book: 2003 Report of the Committee on Infectious Diseases. 28th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2003:237–238.
- Harkinezhad T, Verminnen K, De Buyzere M, Rietzschel E, Bekaert S, Vanrompay, D. Prevalence of Chlamydophila psittaci infections in a human population in contact with domestic and companion birds. J Med Microbiol. 2009;58(pt 9):1207–1212.
- Heddema E, van Hannen EJ, Duim B, et al. An outbreak of psittacosis due to Chlamydophila psittaci genotype A in a veterinary teaching hospital. J Med Microbiol. 2006;55(pt 11):1571–1575.
- Kaibu H, Iida K, Ueki S, et al. Psittacosis in all four members of a family in Nagasaki, Japan. Jpn J Infect Dis. 2006;59(5):349–350.
- Smith KA, Bradley KK, Stobierski MG, Tengelsen LA; National Association of State Public Health Veterinarians Psittacosis Compendium Committee. Compendium of measures to control Chlamydophila psittaci (formerly Chlamydia psittaci) infection among humans (psittacosis) and pet birds, 2005. J Am Vet Med Assoc. 2005;226(4):532–539.
- Geigenfeind I, Haag-Wackernagel D. Detection of Chlamydophila psittaci from feral pigeons in environmental samples: problems with currently available techniques. Integr Zool. 2010;5(1):63–69.
- Petrovay F, Balla E. Two fatal cases of psittacosis caused by Chlamydophila psittaci. J Med Microbiol. 2008;57(pt 10):1296–1298.
- Evans EE. Zoonotic diseases of common pet birds: psittacine, passerine, and columbiform species. Vet Clin North Am Exot Anim Pract. 2011;14(3):457–476.
- Harkinezhad T, Verminnen K, Van Droogenbroeck C, Vanrompay D. Chlamydophila psittaci genotype E/B transmission from African grey parrots to humans. J Med Microbiol. 2007;56(pt 8):1097–1100.
- Matsui T, Nakashima K, Ohyama T, et al. An outbreak of psittacosis in a bird park in Japan. Epidemiol Infect. 2008;136(4):492–495.
- Centers for Disease Control and Prevention. National Notifiable Diseases Surveillance System (NNDSS). Psittacosis/ornithosis (Chlamydophila psittaci) 2010 case definition. https://wwwn.cdc.gov/nndss/conditions/psittacosis/case-definition/2010/. Accessed 25 Jan. 2019.
- Schlossberg D. Psittacosis (due to Chlamydia psittaci). In: Bennett J, Dolin R, Blaser MJ, eds. Mandell, Douglas, and Bennett’s Principles and Practice of Infectious Diseases. 8th ed. Philadelphia, PA: Elsevier/Saunders. 2015:2171–2173.
- Beeckman DS, Vanrompay DC. Zoonotic Chlamydophila psittaci infections from a clinical perspective. Clin Microbiol Infect. 2009;15(1):11–17.
- Persson K, Boman J. Comparison of five serologic tests for diagnosis of acute infections by Chlamydia pneumoniae. Clin Diagn Lab Immunol. 2000;7(5):739–744.
- Bourke SJ, Carrington D, Frew CE, Stevenson RD, Banham SW. Serological cross-reactivity among chlamydial strains in a family outbreak of psittacosis. J Infect. 1989;19(1):41–45.
- Mandell LA, Wunderink RG, Anzueto A, et al. Infectious Diseases Society of America/American Thoracic Society consensus guidelines on the management of community-acquired pneumonia in adults. Clin Infect Dis. 2007;44(suppl 2):S27–S72.
- Elliott JH. Psittacosis: a flu-like syndrome. Aust Fam Physician. 2001;30(8):739–741.
- American Academy of Pediatrics. Tetracyclines. In: Kimberlin DW, Brady MT, Jackson MA, Long SS, eds. Red Book: 2015 Report of the Committee on Infectious Diseases. 30th ed. Elk Grove Village, IL: American Academy of Pediatrics; 2015:873.
- Armed Forces Health Surveillance Branch in collaboration with the U.S. Air Force School of Aerospace Medicine, Army Public Health Center, and Navy and Marine Corps Public Health Center. Armed Forces reportable medical events: guidelines and case definitions. https://health.mil/Reference-Center/Publications/2017/07/17/Armed-Forces-Reportable-Medical-Events-Guidelines. Published 17 July 2017. Accessed 25 Jan. 2019.