Abstract
Lyme disease diagnostic workups conducted on active and retired U.S. service members and their dependents at U.S. Air Force military treatment facilities (MTFs) in Germany between 2013 and 2017 were assessed to determine the appropriateness of laboratory testing and antibiotic prescriptions. Of the 1,176 first-tier immunoassays, 1,114 (94.7%) were negative, and of the 285 immunoglobulin M (IgM) immunoblots, 242 (84.9%) followed a negative first-tier assay or were performed without an antecedent first-tier assay. Eighty-three positive IgM immunoblot tests were adjudicated using modified published criteria, of which 40 (48.2%) were deemed false positives. Thirty-two patients with false-positive tests were treated with an antibiotic. Additionally, 30 patients with uncomplicated erythema migrans could have been treated without laboratory confirmation. Understanding the use and limitations of 2-tier diagnostic criteria, as well as the common pitfalls in diagnosing Lyme disease, may help prevent overdiagnosis, reduce unnecessary testing, and promote antibiotic stewardship.
What Are the New Findings?
Of the 83 positive Lyme disease IgM immunoblots conducted at U.S. Air Force MTF laboratories in Germany between 2013 and 2017, 40 (48.2%) were deemed false positives after standardized chart review, and 32 of these patients were prescribed antibiotics. Thirty patients with true-positive IgM immunoblots could have been diagnosed and treated without laboratory testing.
What Is the Impact on Readiness and Force Health Protection?
Lyme disease is the most commonly diagnosed vector-borne illness in the U.S. military. Early diagnosis and treatment are essential to prevent complications from disseminated disease. Overreliance on serologic testing, given its low positive predictive value in certain contexts, can lead to misdiagnosis, wasted expenditure, and antibiotic misuse.
Background
Lyme disease, known commonly outside the U.S. as Lyme borreliosis, is caused by infection with tick-borne spirochetes of the Borrelia burgdorferi sensu lato complex.1 Clinicians trained in the U.S. who practice internationally or who commonly treat international travelers should understand the universal commonalities and region-specific differences in the microbiology, presentation, and diagnosis of Lyme disease.
In North America, the vast majority of Lyme disease is caused by B. burgdorferi sensu stricto,1 although other presumably pathogenic genospecies have been isolated.2–5 Pathogen diversity is greater in Europe, where B. burgdorferi sensu stricto, B. afzelii, B. garinii, B. bavariensis, and B. spielmanii are established contributors to the burden of human disease. Regardless of the infecting genospecies, Lyme disease can be classified into 3 stages: 1) early localized disease, which occurs days to weeks after the vector tick bite and is often characterized by erythema migrans (i.e., an expanding erythematous skin lesion that may develop central clearing); 2) early disseminated disease, which can follow weeks to months after untreated infection and may present as multiple erythema migrans, Lyme carditis, or neuroborreliosis; and 3) late disease, which may follow months or years after untreated infection and may include arthritis and other dermatologic and neurologic manifestations.1 Although erythema migrans is a frequent manifestation of Lyme disease worldwide, less common clinical syndromes are geographically heterogeneous. Lyme neuroborreliosis, acrodermatitis chronica atrophicans, and borrelial lymphocytoma, for example, are mostly restricted to Europe.1,6
This pattern of universal commonality and regional disparity also applies to the diagnostic workup. Irrespective of location, laboratory testing should be reserved for patients with an intermediate pre-test probability of disease.6–8 Testing parameters, however, should follow local guidelines. While 2-tier serologic testing for Lyme disease is the standard of care in the U.S.,7 Germany,9 and some other parts of Europe,6,8,10 immunoblot band interpretation differs between North America11 and Europe because of variable surface protein expression among the genospecies.12,13 Two-tier testing is usually conducted in a "reflex" manner such that positive or equivocal results on a first-tier immunoassay are followed by the automatic performance of a Western immunoblot to test for immunoglobulin M (IgM) and immunoglobulin G (IgG) antibodies.7
As in the U.S., Lyme disease has attracted much attention in Europe, where guidelines warn of public misconceptions6 and unwarranted testing.8 The extent of overdiagnosis in the U.S. has been documented in endemic areas14–16 and in a military population spanning endemic and non-endemic areas,17 but similar estimates are not available internationally. This study sought to assess the appropriateness of diagnostic workups and treatments of current and retired U.S. service members and their dependents accessing health care services in Germany. The location was chosen because of its high reported incidence of Lyme disease and its sizable population of U.S. military personnel and beneficiaries.1,18,19
Methods
All Lyme disease serologic tests ordered on U.S. service members, military retirees, and their dependent relatives at U.S. Air Force military treatment facilities (MTFs) between 1 Jan. 2013 and 31 Dec. 2017 were retrieved by querying the Composite Health Care System for the key words "Lyme disease" or "B. burgdorferi." Molecular and C6 peptide tests, as well as tests of non-serum samples (e.g., of cerebral spinal fluid), were excluded. The Defense Medical Information System identifier was used to restrict to tests ordered in Germany. Serologic tests were stratified as either first-tier immunoassays (enzyme immunoassays and indirect immunofluorescence assays, which are indistinguishable in the database) or second-tier Western immunoblots. Immunoblots were further classified as IgM or IgG.
For all patients with a positive IgM immunoblot, data were abstracted from the Armed Forces Health Longitudinal Technology Application and the Health Artifact and Image Management Solution by the principal investigator (BJW). All notes within 1 year of the index Lyme disease IgM immunoblot were reviewed to obtain the following information: patient sex and age at presentation; chief complaint or complaints for health care seeking; symptom onset date; laboratory sample collection date; documented travel within 30 days of clinical presentation; reported tick bite; antibiotic prescription(s) for an indication of Lyme disease; and the presence or absence of erythema migrans, acute febrile illness, cranial nerve palsy, carditis, and meningitis. Criteria and diagnostic codes associated with these 5 conditions have been published elsewhere.17 Patients were assumed to have no travel history if none was documented in the chart. Given the potential for prolonged IgM seropositivity,1 the analysis was restricted to a patient's first positive IgM immunoblot during the study period. Cases with insufficient documentation (i.e., had no clinical notes associated with Lyme disease) or patients with no evidence of residing in or traveling to Germany within 30 days of the test were excluded (this may occur if a specimen is shipped to an MTF laboratory in Germany for testing).
Methodology published by Seriburi and colleagues14 was modified to adjudicate positive IgM immunoblots as true or false positives. Positive immunoblot tests were considered true positives unless 1 or more of the following criteria applied: 1) a first-tier test had been omitted or was negative or the time since symptom onset exceeded 30 days with a negative IgG immunoblot; 2) the patient was tested between Dec. and March, when incident infection in Germany is exceedingly rare;19 and/or 3) the patient was asymptomatic or reported only non-specific symptoms. The immunoblot band criterion of Seriburi was not applied because of different banding patterns for pathogenic B. burgdorferi sensu lato genospecies in Germany.
The Armed Forces Disease Reporting System internet (DRSi) was queried to determine if the IgM positive cases were reported between 1 Jan. 2013 and 30 Sept. 2018 (to account for delayed reporting up to 9 months). All Lyme disease cases diagnosed at MTFs must be reported electronically to this system.20 Based on U.S. Council of State and Territorial Epidemiologists definitions, cases are classified by local public health authorities as suspected, probable, or confirmed.21
Descriptive statistics and 2-sided Fisher exact tests with 95% confidence intervals (CIs) were used to describe the history and clinical presentation of patients and to compare false-positive proportions by sex and by age (children [aged <18 years] versus adults [aged =18 years]). Data were analyzed using SAS/STAT® software, version 9.4 (2014, SAS Institute, Cary, NC). The study was approved by the Air Force Research Laboratory Institutional Review Board.
Results
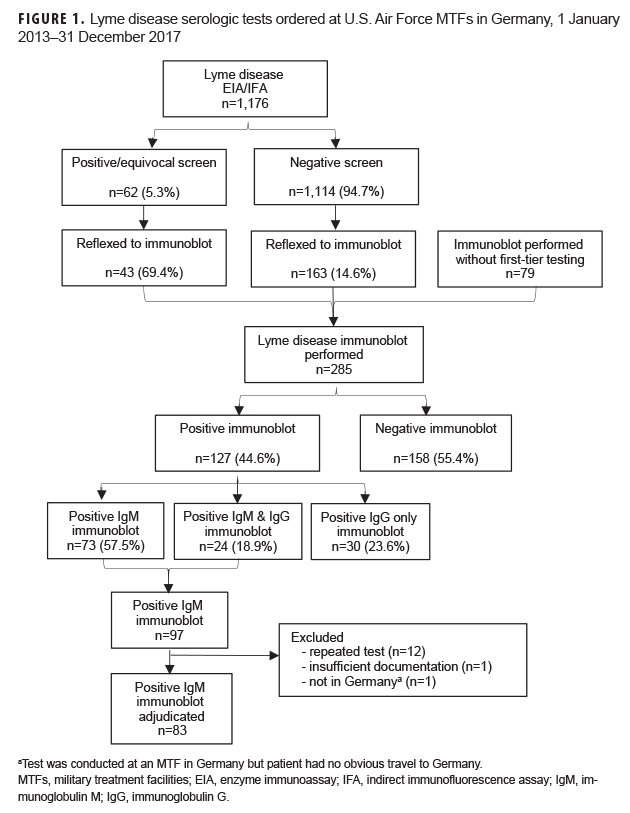
A total of 1,461 serum tests (1,176 immunoassays and 285 immunoblots) were performed on 1,026 unique patients. The mean age of tested patients was 33 years (range: 2 months to 87 years), and 53.8% were female. Of the 1,176 first-tier serologic tests, 62 (5.3%) were positive or equivocal. Of these 62 positive or equivocal first-tier assays, 43 (69.4%) were reflexed to immunoblotting. Seventy-nine additional immunoblots were performed without first-tier testing, and 163 were performed after a negative screen. Of the 285 immunoblots performed, 127 (44.6%) were positive: IgM only (n=73); IgG only (n=30); IgM and IgG (n=24). Fourteen of the 97 positive IgM tests were excluded, leaving 83 cases available for adjudication (Figure 1).
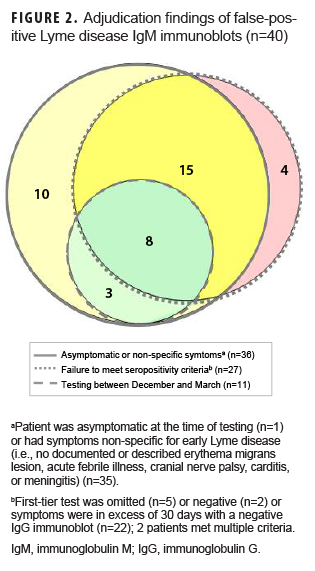
Of the 83 positive IgM immunoblots, 43 (51.8%; 95% CI: 40.6–62.9) were deemed true positives and 40 (48.2%; 95% CI: 37.1–59.4) were deemed false positives. The most common false-positive criterion was asymptomatic or non-specific presentation (n=36), followed by failure to meet seropositivity criteria (n=27). Eleven patients were tested in Dec. through March, but all met at least 1 other false-positive criterion (Figure 2). Among the 83 persons with positive IgM immunoblots, false-positive proportions differed by age (adults [36/63] and children [4/20]; p=.007) and by sex (females [26/40] and males [14/43]; p=.006).
Clinical presentation of the 43 patients with true-positive tests included erythema migrans (n=30), acute febrile illness (n=9), facial palsy (n=2), and carditis (n=2) (data not shown). A tick bite was reported by 19 (44.2%) patients with a true-positive test and 5 (12.5%) patients with a false-positive test. Among the 40 persons who were deemed to have false-positive tests, 1 patient was asymptomatic at presentation, and 35 presented with a variety of chief complaints: arthralgia (n=14), non-erythema migrans skin rash (n=10), headache (n=5), fatigue (n=3), neuropathy (n=3), movement disorder (n=2), myalgia (n=2), abscess (n=1), and cough (n=1); some patients had more than 1 chief complaint (data not shown).
Antibiotics were prescribed for Lyme disease for 41 (95.3%) patients with a true positive test. This included oral doxycycline (n=27), oral amoxicillin (n=11), intravenous ceftriaxone (n=2), and oral erythromycin (n=1). Thirty-two (80.0%) patients with a false-positive test were prescribed antibiotics: oral doxycycline (n=27), oral amoxicillin (n=4), and oral cefuroxime (n=1) (data not shown).
Thirty-six of the 83 positive IgM immunoblot cases were reported as Lyme disease to the DRSi, including 21/43 (48.8%) patients with true-positive tests and 15/40 (37.5%) with false-positive tests. Eighteen patients with true-positive tests were classified as confirmed compared to 13 patients with false-positive tests (data not shown).
Editorial Comment
Nearly 95% of the 1,176 Lyme disease immunoassays ordered at U.S. Air Force MTFs in Germany were negative. Of the 83 positive IgM immunoblot tests reviewed, 43 were adjudicated as true positives and 40 as false positives. Thirty-two (80.0%) patients with false-positive tests were treated with antibiotics.
These data provide valuable information for military clinicians stationed in Germany. First, this study suggests Lyme disease serologic testing is overutilized at MTFs. Only 5.3% of the first-tier immunoassays performed during the surveillance period were positive or equivocal, well below the 11.9% positivity observed at U.S. commercial laboratories (n=287,595 tests).22 Therefore, imperfect sensitivity of Lyme disease serology does not fully explain the large number of negative results in this study. A more likely cause is the use of serology in the workup of vague or non-specific symptoms (e.g., arthralgia, headache, or fatigue).1 While these symptoms may occur during early Lyme disease, they are highly prevalent across the population.7 In the absence of objective findings, Lyme disease serologic testing in these cases is discouraged.7,8,23 Subjective arthralgia of early Lyme disease is clinically distinguishable from Lyme arthritis, a potential manifestation of late disease. The latter affects large joints, presents with objective synovitis, and is almost always pauciarticular (and usually monoarticular). For patients who present with potential Lyme arthritis, the diagnostic workup may include serologic testing and PCR testing of synovial fluid.6,7
Second, and relatedly, this study indicates overreliance on laboratory testing to diagnose Lyme disease. In endemic areas, such as much of Germany,19 patients who present with the typical erythema migrans lesion should be diagnosed and treated without awaiting the results of laboratory testing. This recommendation is based on the recognition that the vast majority of patients with erythema migrans will eventually test positive for IgM antibody but that such serologic tests are usually negative in the early stages of infection.7,8 In this study, the 30 patients with erythema migrans represented 36.1% of the 83 patients with positive IgM immunoblots and 69.8% of the 43 patients determined to have true-positive IgM immunoblots. In the U.S.7 and Europe,9 serologic testing is recommended when the dermatologic presentation is unclear or if other manifestations, such as borrelial lymphocytoma, are suspected. Testing in Germany should follow a 2-tier serologic approach10 guided by specific European immunoblot band interpretation,13 with supplemental molecular testing for certain manifestations, as suggested by European guidelines.8,10,24
Third, this study highlights the issue of antibiotic misuse. Clinicians are encouraged to prescribe antibiotics judiciously. Of the 40 patients with false-positive tests, 32 were provided antibiotics for the indication of Lyme disease. Like superfluous laboratory testing, unnecessary antibiotic utilization is a wasteful expenditure. Moreover, it may lead to complications and encourage antimicrobial resistance.1
Fourth, this study uncovers potential mismanagement of immunoassays. According to universal recommendations regarding 2-tier testing for Lyme disease,6–10 positive or equivocal immunoassays should be reflexively referred for immunoblotting. This occurred for only 43/62 (69.4%) eligible immunoassays. Meanwhile, 163/1,114 (14.6%) negative immunoassays were reflexed to immunoblots, and 79 immunoblots were performed without a first-tier immunoassay. Therefore, of the 285 immunoblots performed, 242 (84.9%) were not in accordance with recommendations. Laboratories conducting Lyme disease testing may consider modifying their processes.
This study has several limitations. First, it relies on serology for ascertainment of potential cases. Serologic diagnosis of Lyme disease is problematic because of imperfect sensitivity and specificity, methodological discrepancies between laboratories, and subjective interpretation of immunoblot banding patterns.25 Second, incomplete or inaccurate data in the medical charts may have resulted in differential misclassification of cases. For example, provider failure to detect, describe, or diagnose the presence of an erythema migrans lesion would result in a false-positive misclassification. Conversely, underestimation of symptom duration in a patient with a negative IgG would result in a true-positive misclassification. Third, no data were collected on patients who had negative tests. Although the low percentage of positive immunoassays suggests a suboptimal pretest prevalence of disease in the tested population,1,12 the appropriateness of ordering these tests could not be assessed. Fourth, patients included in this study were predominantly evaluated at U.S. air bases in southwest Germany. Given the intranational heterogeneity of Lyme disease,19 the findings may not be generalizable to all U.S. service members, military retirees, and their dependents stationed or residing in Germany.
Lyme disease is the most commonly reported tick-borne disease in Germany19and the most commonly reported vector-borne disease in the U.S. Armed Forces.26 Two air bases in Germany account for 16.4% of all vector-borne diseases reported in the U.S. Air Force.18The present study highlights clinical challenges associated with Lyme disease and demonstrates their applicability outside the U.S.23Military clinicians practicing anywhere in Germany should understand these challenges and recognize that patients may access online information that is often inaccurate.27High-quality, evidence-based care may include diagnosing and treating Lyme disease without laboratory testing, explaining why laboratory testing is unwarranted for non-specific symptoms, and practicing good antibiotic stewardship.
Author Affiliations: School of Medicine, Creighton University, Omaha, NE (2d Lt Nelson); Public Health and Preventive Medicine Department, U.S. Air Force School of Aerospace Medicine, Wright-Patterson AFB, OH (Ms. Burganowski, Dr. Colton, Mr. Escobar, Lt Col Gambino-Shirley, Maj Webber); Oak Ridge Institute for Science & Education, Department of Energy, Oak Ridge, TN (Ms. Burganowski); DataRev LLC, Atlanta, GA (Ms. Pathak); Solutions Through Innovative Technologies, Inc., Fairborn, OH (Ms. Pathak)
Acknowledgements: The authors wish to thank Ms. Sandy Kawano, technical editor, Aeromedical Research Department, U.S. Air Force School of Aerospace Medicine, for her assistance editing this manuscript. This project was supported in part by an appointment to the Research Participation Program for the U.S. Air Force School of Aerospace Medicine, Public Health and Preventive Medicine Department, administered by the Oak Ridge Institute for Science and Education through an agreement between the U.S. Department of Energy and U.S. Air Force School of Aerospace Medicine, Public Health and Preventive Medicine Department. The views expressed in this article are those of the authors and do not necessarily reflect the official policy or position of the Air Force, the Department of Defense, or the U.S. Government.
References
- Stanek G, Wormser GP, Gray J, Strle F. Lyme borreliosis. Lancet. 2012;379:461–473.
- Pritt BS, Mead PS, Johnson DKH, et al. Identification of a novel pathogenic Borrelia species causing Lyme borreliosis with unusually high spirochaetaemia: a descriptive study. Lancet Infect Dis. 2016;16(5):556–564.
- Clark KL, Leydet B, Hartman S. Lyme borreliosis in human patients in Florida and Georgia, USA. Int J Med Sci. 2013;10(7):915–931.
- Girard YA, Fedorova N, Lane RS. Genetic diversity of Borrelia burgdorferi and detection of B. bissettii-like DNA in serum of north-coastal California residents. J Clin Microbiol. 2011;49(3):945– 954.
- Caporale DA, Johnson CM, Millard BJ. Presence of Borrelia burgdorferi (Spirochaetales: Spirochaetaceae) in southern Kettle Moraine State Forest, Wisconsin, and characterization of strain W97F51. J Med Entomol. 2005;42(3):457–472.
- Stanek G, Fingerle V, Hunfeld KP, et al. Lyme borreliosis: Clinical case definitions for diagnosis and management in Europe. Clin Microbiol Infect. 2011;17(1):69–79.
- Wormser GP, Dattwyler RJ, Shapiro ED, et al. The clinical assessment, treatment, and prevention of Lyme disease, human granulocytic anaplasmosis, and babesiosis: clinical practice guidelines by the Infectious Diseases Society of America. Clin Infect Dis. 2006;43(9):1089–1134.
- Dessau RB, van Dam AP, Fingerle V, et al. To test or not to test? Laboratory support for the diagnosis of Lyme borreliosis: a position paper of ESGBOR, the ESCMID study group for Lyme borreliosis. Clin Microbiol Infect. 2018;24(2):118–124.
- Hofmann H, Fingerle V, Hunfeld KP, et al. Cutaneous Lyme borreliosis: guideline of the German Dermatology Society. Ger Med Sci. 2017;15:Doc14.
- Brouqui P, Bacellar F, Baranton G, et al. Guidelines for the diagnosis of tick-borne bacterial diseases in Europe. Clin Microbiol Infect. 2004;10(12):1108–1132.
- Centers for Disease Control and Prevention. Recommendations for test performance and interpretation from the Second National Conference on Serologic Diagnosis of Lyme Disease. MMWR Morb Mortal Wkly Rep. 1995;44(31):590–591.
- Robertson J, Guy E, Andrews N, et al. A European multicenter study of immunoblotting in serodiagnosis of Lyme disease. J Clin Microbiol. 2000;38(6):2097–2102.
- Hauser U, Lehnert G, Lobentanzer R, Wilske B. Interpretation criteria for standardized Western blots for three European species of Borrelia burgdorferi sensu lato. J Clin Microbiol. 1997;35(6):1433–1444.
- Seriburi V, Ndukwe N, Chang Z, Cox ME, Wormser GP. High frequency of false positive IgM immunoblots for Borrelia burgdorferi in clinical practice. Clin Microbiol Infect. 2012;18(12):1236–1240.
- Lantos PM, Lipsett SC, Nigrovic LE. False positive Lyme disease IgM immunoblots in children. J Pediatr. 2016;174:267–269.
- Lantos PM, Branda JA, Boggan JC, et al. Poor positive predictive value of Lyme disease serologic testing in an area of low disease incidence. Clin Infect Dis. 2015;61(9):1374–1380.
- Webber BJ, Burganowski RP, Colton L, Escobar JD, Pathak SR, Gambino-Shirley KJ. Lyme disease overdiagnosis in a large health care system: a population-based, retrospective study. Clin Microbiol Infect. 2019;S1198-743X(19)30086-2. doi:10.1016/j.cmi.2019.02.020.
- Anna MM, Escobar JD, Chapman AS. Reported vector-borne and zoonotic diseases, U.S. Air Force, 2000–2011. MSMR. 2012;19(10):11–14.
- Enkelmann J, Böhmer M, Fingerle V, et al. Incidence of notified Lyme borreliosis in Germany, 2013–2017. Sci Rep. 2018;8:14976.
- Defense Health Agency. Armed Forces Health Surveillance Branch. Armed Forces Reportable Medical Events. Guidelines and Case Definitions. https://health.mil/REFERENCE-CENTER/PUBLICATIONS/2017/07/17/ARMED-FORCES-REPORTABLE-MEDICAL-EVENTS-GUIDELINES. Accessed 19 July 2019.
- Centers for Disease Control and Prevention. Lyme disease (Borrelia burgdorferi): 2017 case definition. https://wwwn.cdc.gov/nndss/conditions/lyme-disease/case-definition/2017/. Accessed 19 July 2019.
- Hinckley AF, Connally NP, Meek JI, et al. Lyme disease testing by large commercial laboratories in the United States. Clin Infect Dis. 2014;59(5):676–681.
- Moore A, Nelson C, Molins C, Mead P, Schriefer M. Current guidelines, common clinical pitfalls, and future directions for laboratory diagnosis of Lyme disease, United States. Emerg Infect Dis. 2016;22(7):1169–1177.
- Mygland A, Ljøstad U, Fingerle V, Rupprecht T, Schmutzhard E, Steiner I. EFNS guidelines on the diagnosis and management of European Lyme neruoborreliosis. Eur J Neurol. 2010;17(1):8–16.
- Aguero-Rosenfeld ME, Wormser GP. Lyme disease: diagnostic issues and controversies. Expert Rev Mol Diagn. 2015;15(1):1–4.
- O’Donnell FL, Stahlman S, Fan M. Surveillance for vector-borne diseases among active and reserve component service members, U.S. Armed Forces, 2010–2016. MSMR. 2018;25(2):8–15.
- Cooper JD, Feder HM Jr. Inaccurate information about Lyme disease on the Internet. Pediatr Infect Dis J. 2004;23(12):1105–1108. Credit: Centers